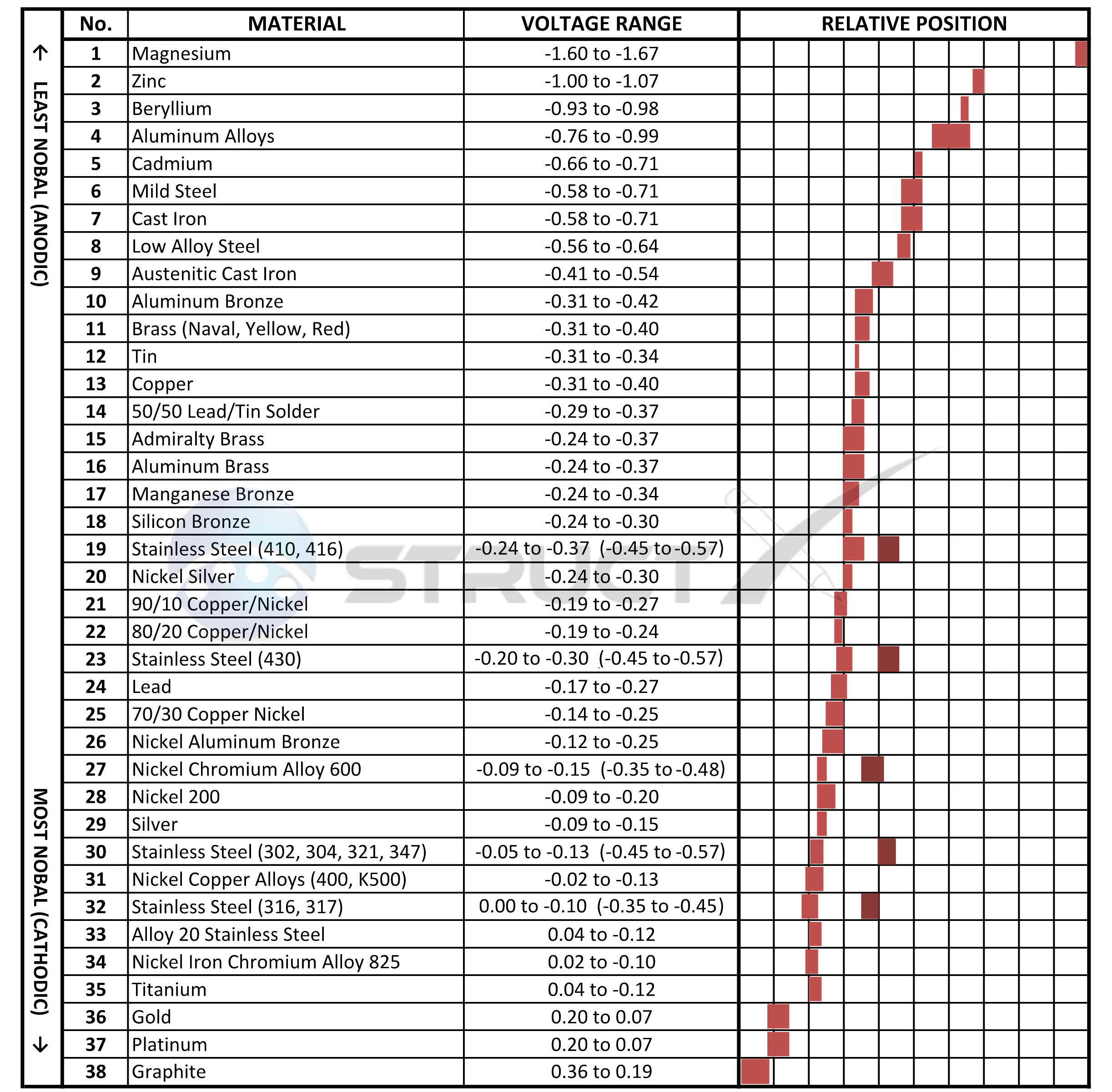
Galvanic Series (Scale of Nobility)
Galvanic Corrosion
Whenever different metals are positioned in an electrolyte within close proximity to one another preferential corrosion becomes an important factor to be aware of. Galvanic corrosion typically attacks junction areas of dissimilar metals and occurs when the following three conditions are met.
- Condition 1: Metals must have a galvanic incompatibility (voltage difference - see below table).
- Condition 2: An electrolyte must be present (water is a good example).
- Condition 3: There must be an electrical conducting path between the metals.
The galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). A typical rule of thumb is that voltage differences of 0.2 Volts or more suggest a galvanic corrosion risk. Generally speaking the further apart the metals in the galvanic series the greater the risk of corrosion, with the metal being of least noble (Anodic) class typically corroding.
It should be noted that although stainless steels are generally in the more noble or cathodic region in the series, studies have shown that stainless steels exhibit a different voltage range in crevices or stagnant and poorly aerated water (these values are shown in brackets).
Galvanic Corrosion Risk
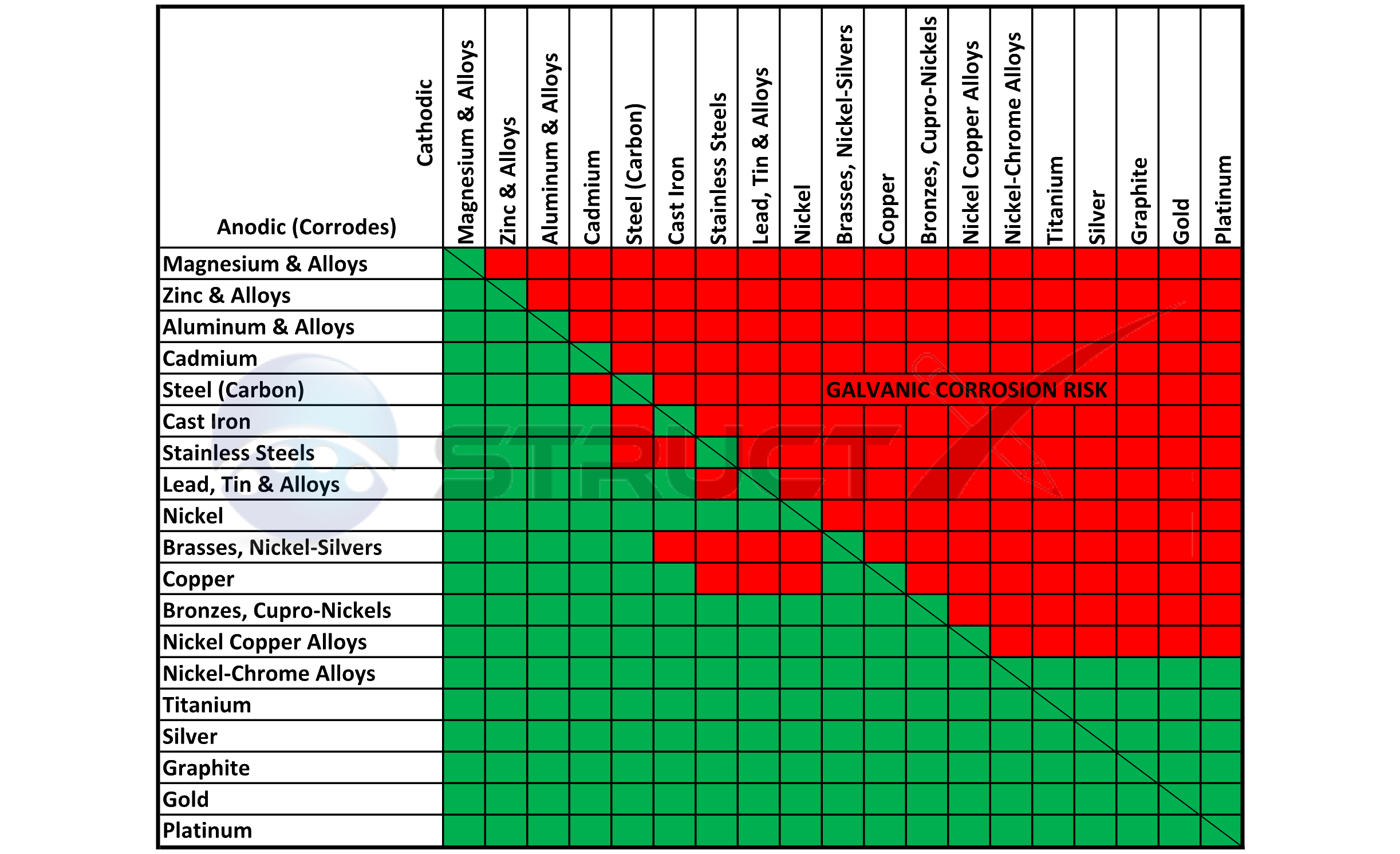
The below table can be used as a quick guide to select compatible metals. Where two materials meet in a green region the corrosion potential can be thought of negligible, meaning, a good combination. Materials that meet in the red zone however should be avoided or used with caution.
Additional Resources
- C. Houska. Stainless Steels in Architecture, Building and Construction: Guidelines for Corrosion Prevention. Nickel Institute, USA. 1965. Publised article.
- Euro Inox. Stainsless Steel in Contact with Other Metallic Materials, volume 10. Belgium. 2005. Print.
- R. Baboian. R.S. Treseder. Nace Corrosion Engineer's Book. Texas. 2002. Print.
- ResearchGate. (2014, March). Galvanic series of various materials in flowing seawater (2.5-4 m/s) at temperatures in the range from 5 to 30oC. [ upload Retrieved from https://www.researchgate.net/figure/Galvanic-series-of-various-materials-in-flowing-seawater-25-4-m-s-at-temperatures-in_fig1_260528991
